|
by Maximilian EMMERT, Theo KOFIDIS and Uwe KLIMA
 he heart has always been regarded as a terminally differentiated organ without any capacity of regeneration after damage. This central dogma which lasted 50 years has been challenged recently. Since a few years ago, the application of stem cells for the heart is undergoing evaluation. The idea of using the own cells of the body has raised new hope for myocardial restoration and may be a breakthrough for future cardiac therapy. he heart has always been regarded as a terminally differentiated organ without any capacity of regeneration after damage. This central dogma which lasted 50 years has been challenged recently. Since a few years ago, the application of stem cells for the heart is undergoing evaluation. The idea of using the own cells of the body has raised new hope for myocardial restoration and may be a breakthrough for future cardiac therapy.
R&D Goals
The concept of using stem cells for cardiac repair could be a feasible solution for this problem and might open new pathways of cardiac therapy in the future. Therefore, different stem cell populations are undergoing evaluation with a special focus on their regenerative potential as well as the most effective way of their clinical application. The cell source that is mainly used is still the autologous bone marrow, which is home to various cell types including hematopoetic stem cells, mesenchymal stem cells, endothelial progenitors and the so-called side population cells (SP cells). Further, myoblasts as well as embryonic stem cells are suggested for myocardial repair. The routes of stem cell delivery that are mainly suggested are the intra-coronary infusion and the intra-myocardial injection, either catheter-based or during surgery.
 However, despite all these various cell types, the success of stem cell-based therapies is still not satisfying and the introduction into the clinical setting is very limited. The main causes for these problems are the lack of effectiveness, non-specific differentiation and insufficient cell amounts for large-scale restoration. In addition, severe safety issues further complicate the clinical usage. For example, in case of myoblast transfer, life-threatening arrhythmia has been reported as these cells do not couple with the host cardiomyocytes after transplantation. However, despite all these various cell types, the success of stem cell-based therapies is still not satisfying and the introduction into the clinical setting is very limited. The main causes for these problems are the lack of effectiveness, non-specific differentiation and insufficient cell amounts for large-scale restoration. In addition, severe safety issues further complicate the clinical usage. For example, in case of myoblast transfer, life-threatening arrhythmia has been reported as these cells do not couple with the host cardiomyocytes after transplantation.
New hopes to solve these problems arose when evidences accumulated recently that the heart is host to its own stem cell populations with the capacity to differentiate towards all cardiac cell lineages. These findings were followed by characterization and even isolation of cardiac resident stem cells (CRSCs). Researchers from several laboratories identified stem cell markers which are specific for cell populations residing in the heart mainly including cells expressing C-kit (CD 117, stem cell factor receptor), side population cells expressing BCRP (breast cancer resistance protein / ABCG2) and Sca-1+ (stem cell antigen-1) cells. These stem cell populations are considered to hold specific regenerative potential with the capacity to differentiate into cardiomyocytes, smooth muscle cells as well as endothelial cells. For this reason, they are suggested as a promising cell source to be used for myocardial restoration in the future.
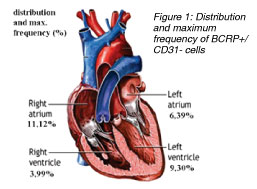
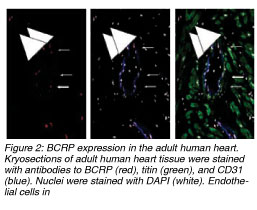
In one of our current studies, we identified and quantified C-kit+ and BCRP+ (ABCG2) cardiac resident stem cells in ischemic and non-ischemic human heart. Further, we performed mapping of the heart for these cell populations as the detailed knowledge of distribution and action of CRSCs before and after ischemic episodes might have an important impact on future stem cell treatment strategies. We obtained 55 biopsies from 50 patients during heart surgery from atria and ventricles and performed staining for BCRP (breast cancer resistance protein / ABCG2, representing the side population phenotype) and C-kit (CD 117, stem cell factor receptor). The BCRP+ cells were excluded from the microscopic field cell count whenever they co-stained for CD-31 (endothelial cell marker). Similarly, the C-kit+ cells which stained positive for mast cell tryptase were excluded too. A titin stain was used to identify stem cells with a cardiac phenotype.]]
We identified BCRP+/CD31- cells which represent cardiac resident stem cells in all areas of the human heart. The highest frequency of BCRP+/CD31- cells was detected in the ischemic tissue of the right atria with a maximum of 11.10% vs. 7.75% (5.62% ¡À 2.51% vs. 4.33% ¡À 2.52%) in non-ischemic atria. Further, a higher amount of BCRP+/CD31- cells was found in the ischemic ventricles within and around the area of an infarction (5.73% ¡À 2.39% vs. 1.39% ¡À 1.79%) compared to healthy myocardium. In 50% of the samples, a low number of BCRP+ cells co-expressed Titin. Also, C-kit+ cells were found in higher numbers within injured (1: 25,000 of cell counts) vs. healthy myocardium (1:105,000). The C-kit+ cells seemed round-shaped, small, and did not stain for cardiac marker Titin. We did not find double staining in BCRP+/C-kit+ cells.
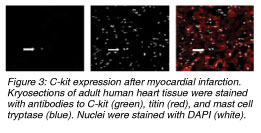 In compliance with several studies conducted before, our results show that the human heart holds self-renewing potential and repair mechanisms which are specifically activated after ischemic events. Further, the atria of a human heart contain higher numbers of resident stem cells than the ventricles. And, as very similar levels of these cells were detected in ischemic but also in non-ischemic atrial tissue, the atria may be considered as a reservoir for cardiac resident stem cells. Moreover, our results lead to the important question whether the ventricle hosts its own BCRP+ stem cells which are being activated after ischemic events, or myocardial injury induces the immigration of these cells from the atrial reservoir into the ventricle. As we found very low numbers of BCRP+ in non-ischemic ventricle, the suggestion of stem cell moving from the atrium into the ventricle may be conceivable. From the clinical point of view, the localization of CRSCs in the atria gives an excellent opportunity for a safe and effective isolation during heart surgery or catheterization. Furthermore, we could confirm the presence of C-kit+ cardiac resident stem cells in the adult myocardium, and similar to BCRP+ resident stem cells, they were also detected in higher frequencies in the ischemic ventricles whereas their presence and their total numbers in healthy myocardium were much lower. Finally, we could show that there was no detection of C-kit+ / BCRP+ co-expressing stem cell populations, which leads to the fact that these two markers express two distinct cardiac resident stem cell populations in the human heart. In compliance with several studies conducted before, our results show that the human heart holds self-renewing potential and repair mechanisms which are specifically activated after ischemic events. Further, the atria of a human heart contain higher numbers of resident stem cells than the ventricles. And, as very similar levels of these cells were detected in ischemic but also in non-ischemic atrial tissue, the atria may be considered as a reservoir for cardiac resident stem cells. Moreover, our results lead to the important question whether the ventricle hosts its own BCRP+ stem cells which are being activated after ischemic events, or myocardial injury induces the immigration of these cells from the atrial reservoir into the ventricle. As we found very low numbers of BCRP+ in non-ischemic ventricle, the suggestion of stem cell moving from the atrium into the ventricle may be conceivable. From the clinical point of view, the localization of CRSCs in the atria gives an excellent opportunity for a safe and effective isolation during heart surgery or catheterization. Furthermore, we could confirm the presence of C-kit+ cardiac resident stem cells in the adult myocardium, and similar to BCRP+ resident stem cells, they were also detected in higher frequencies in the ischemic ventricles whereas their presence and their total numbers in healthy myocardium were much lower. Finally, we could show that there was no detection of C-kit+ / BCRP+ co-expressing stem cell populations, which leads to the fact that these two markers express two distinct cardiac resident stem cell populations in the human heart.
|
Objectives: Since the heart has been reported to hold its own stem cell populations, these so-called cardiac resident stem cells (CRSC) might be a breakthrough for myocardial restoration in the future. However, so far these cells have not been studied in detail. In a recent study, we analyzed their frequency and their distribution pattern in biopsies from patients undergoing cardiac surgery.
Methods: We obtained 55 human biopsies from atria and ventricles and performed staining for BCRP (breast cancer resistance protein) and C-kit (CD 117, stem cell factor receptor) which have been described as specific marker proteins for cardiac resident stem cells.
Results: The highest frequency of BCRP+ cells was found in the atria (11% of the total number of cells). A higher amount of BCRP+ cells was found in the ischemic ventricles within and around the area of an infarction compared to healthy myocardium. In addition, C-kit+ cells were also found in higher numbers within injured vs. healthy myocardium.
Conclusions: The atria of a human heart contain a higher number of resident stem cells than the ventricles. Their population is also higher within and around the infarcted areas (post acute infarction). A migration of stem cells to the ventricle is possible as a repair mechanism.
|
Cardiac resident stem cells have raised new hopes for myocardial restoration and after the successful identification of various cell populations, specific steps have to be followed by further investigation. Stem cell dynamics as well as effective cell isolation and expansion techniques to generate adequate cell amounts for clinical application should be the next aims for the future. The suggestion that human atria seem to constitute a reservoir of CRSCs and the evaluation of their distribution pattern before and after ischemic events could have an important impact for a better understanding of stem cell dynamics and cardiac homeostasis. This may be a decisive step for the future development of promising therapy strategies and could hold great potential for future myocardial restoration.
 Click here to download the full issue for USD 6.50 Click here to download the full issue for USD 6.50
|


 he heart has always been regarded as a terminally differentiated organ without any capacity of regeneration after damage. This central dogma which lasted 50 years has been challenged recently. Since a few years ago, the application of stem cells for the heart is undergoing evaluation. The idea of using the own cells of the body has raised new hope for myocardial restoration and may be a breakthrough for future cardiac therapy.
he heart has always been regarded as a terminally differentiated organ without any capacity of regeneration after damage. This central dogma which lasted 50 years has been challenged recently. Since a few years ago, the application of stem cells for the heart is undergoing evaluation. The idea of using the own cells of the body has raised new hope for myocardial restoration and may be a breakthrough for future cardiac therapy.

 However, despite all these various cell types, the success of stem cell-based therapies is still not satisfying and the introduction into the clinical setting is very limited. The main causes for these problems are the lack of effectiveness, non-specific differentiation and insufficient cell amounts for large-scale restoration. In addition, severe safety issues further complicate the clinical usage. For example, in case of myoblast transfer, life-threatening arrhythmia has been reported as these cells do not couple with the host cardiomyocytes after transplantation.
However, despite all these various cell types, the success of stem cell-based therapies is still not satisfying and the introduction into the clinical setting is very limited. The main causes for these problems are the lack of effectiveness, non-specific differentiation and insufficient cell amounts for large-scale restoration. In addition, severe safety issues further complicate the clinical usage. For example, in case of myoblast transfer, life-threatening arrhythmia has been reported as these cells do not couple with the host cardiomyocytes after transplantation.
 In compliance with several studies conducted before, our results show that the human heart holds self-renewing potential and repair mechanisms which are specifically activated after ischemic events. Further, the atria of a human heart contain higher numbers of resident stem cells than the ventricles. And, as very similar levels of these cells were detected in ischemic but also in non-ischemic atrial tissue, the atria may be considered as a reservoir for cardiac resident stem cells. Moreover, our results lead to the important question whether the ventricle hosts its own BCRP+ stem cells which are being activated after ischemic events, or myocardial injury induces the immigration of these cells from the atrial reservoir into the ventricle. As we found very low numbers of BCRP+ in non-ischemic ventricle, the suggestion of stem cell moving from the atrium into the ventricle may be conceivable. From the clinical point of view, the localization of CRSCs in the atria gives an excellent opportunity for a safe and effective isolation during heart surgery or catheterization. Furthermore, we could confirm the presence of C-kit+ cardiac resident stem cells in the adult myocardium, and similar to BCRP+ resident stem cells, they were also detected in higher frequencies in the ischemic ventricles whereas their presence and their total numbers in healthy myocardium were much lower. Finally, we could show that there was no detection of C-kit+ / BCRP+ co-expressing stem cell populations, which leads to the fact that these two markers express two distinct cardiac resident stem cell populations in the human heart.
In compliance with several studies conducted before, our results show that the human heart holds self-renewing potential and repair mechanisms which are specifically activated after ischemic events. Further, the atria of a human heart contain higher numbers of resident stem cells than the ventricles. And, as very similar levels of these cells were detected in ischemic but also in non-ischemic atrial tissue, the atria may be considered as a reservoir for cardiac resident stem cells. Moreover, our results lead to the important question whether the ventricle hosts its own BCRP+ stem cells which are being activated after ischemic events, or myocardial injury induces the immigration of these cells from the atrial reservoir into the ventricle. As we found very low numbers of BCRP+ in non-ischemic ventricle, the suggestion of stem cell moving from the atrium into the ventricle may be conceivable. From the clinical point of view, the localization of CRSCs in the atria gives an excellent opportunity for a safe and effective isolation during heart surgery or catheterization. Furthermore, we could confirm the presence of C-kit+ cardiac resident stem cells in the adult myocardium, and similar to BCRP+ resident stem cells, they were also detected in higher frequencies in the ischemic ventricles whereas their presence and their total numbers in healthy myocardium were much lower. Finally, we could show that there was no detection of C-kit+ / BCRP+ co-expressing stem cell populations, which leads to the fact that these two markers express two distinct cardiac resident stem cell populations in the human heart.
 Click here to download the full issue for USD 6.50
Click here to download the full issue for USD 6.50